
In addition, vinyl ether-Tz, recently introduced by Fox et al., (29) have been shown to exhibit increased IEDDA reactivity despite the non-electron-withdrawing nature of the substituent ( Figure 1b), while also maintaining high stability. (33−35) So far, 4-pyridyl-Tz ( Figure 1b) have only rarely been used, (36) and there is no comparative data on the IEDDA reaction kinetics of 2-pyridyl- and 4-pyridyl-Tz. (2,3,32) Assuming that the reactivity is indeed controlled by frontier molecular orbital (FMO) interactions, we hypothesized that 4-pyridyl-substituted Tz are even more reactive than their 2-pyridyl analogues. This is commonly attributed to the electron-withdrawing effect of the 2-pyridyl substituent, resulting in a lowered orbital energy of the Tz, thereby accelerating the IEDDA cycloaddition ( Figure 1b). (27−31) In particular, 2-pyridyl-Tz are frequently used despite limited stability because of their exceptionally high reactivity. These applications have motivated and fueled the development of advanced procedures for the synthesis of Tz scaffolds. 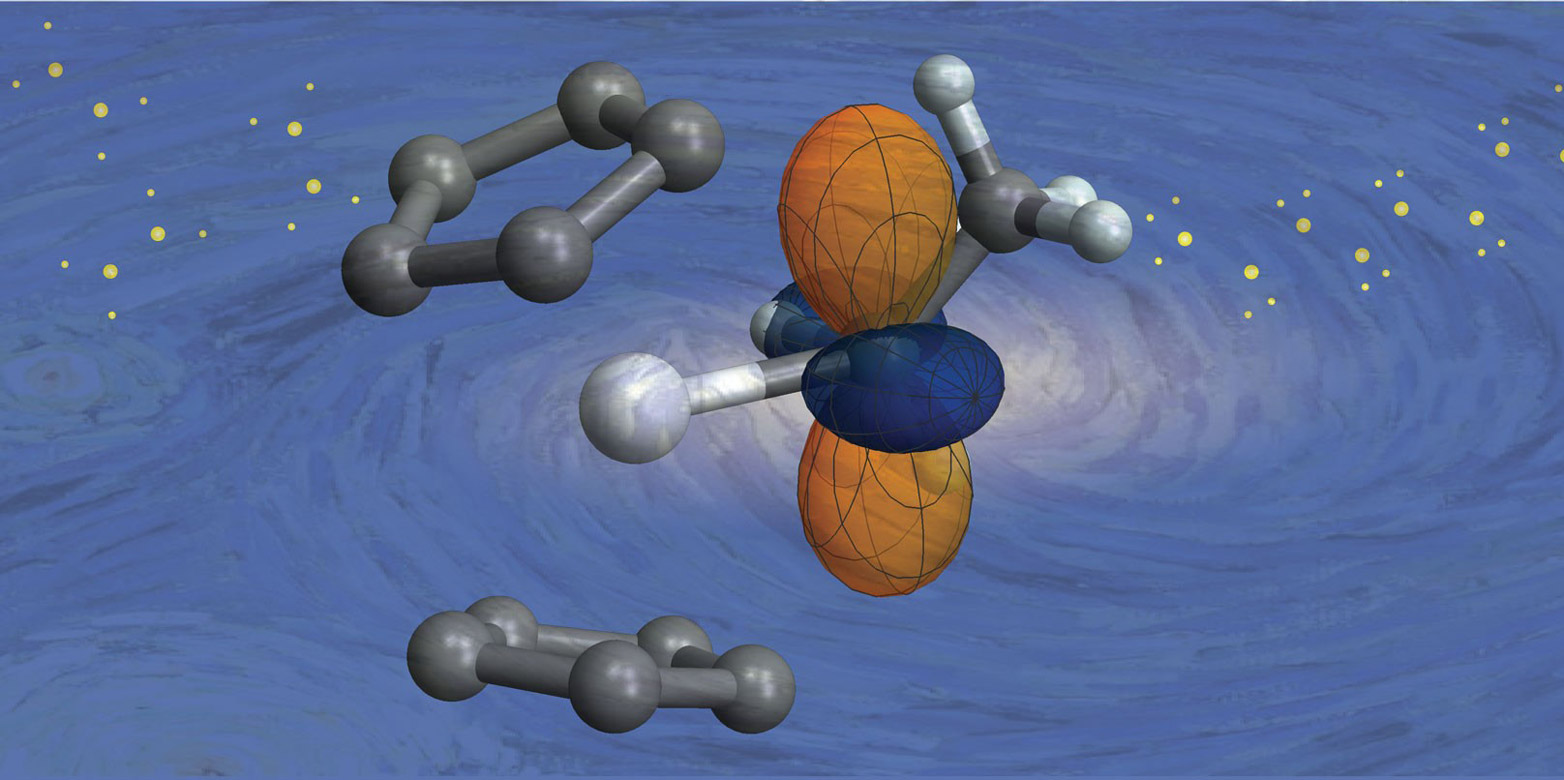
In recent years, a variety of differently substituted Tz have been used for bioorthogonal reactions and in vivo chemistry, including bis-alkyl-substituted Tz, (11−15) alkyl-aryl-Tz, (16−19) mono-alkyl-Tz (20) (alkyl-H-Tz) as well as highly reactive bis-heteroaryl (21,22) and mono-aryl (23−26) derivatives (aryl-H-Tz). (2,3) Very recently, bioorthogonal chemistry has entered phase 1 clinical trials, with tetrazine/ trans-cyclooctene reactions currently being tested in humans, aiming for locally restricted prodrug activation to improve the selectivity of chemotherapeutics. Due to high reaction rates, its biocompatibility, and versatility, the tetrazine/ trans-cyclooctene ligation has found broad applications in many fields, in particular enabling selective chemical reactions in living organisms. (8) In the rate-determining step, the tetrazine first reacts with the trans-cyclooctene in a -cycloaddition reaction to form a bicyclic intermediate that rapidly undergoes cycloreversion to give dihydropyridazines as ligation products ( Figure 1a). (1−3) In particular, trans–cyclooctene ( TCO) derivatives (4−6) provide several orders of magnitude higher reactivity than other dienophiles such as cyclopropenes (7) or norbornenes. The inverse electron demand Diels–Alder (IEDDA)-initiated ligation of 1,2,4,5-tetrazines (Tz) with strained dienophiles represents a group of exceptionally fast bioorthogonal reactions. These fundamental insights reveal thus far overlooked mechanistic aspects that govern the reactivity/stability trade-off for tetrazines in physiologically relevant environments, thereby providing a new strategy that may facilitate the rational design of these bioorthogonal tools.  
We show that the limited stability of tetrazines in biological media strongly correlates with the electron-withdrawing effect of the substituent, while intramolecular repulsion increases the reactivity without reducing the stability. Computational investigation of this phenomenon revealed that distortion of the tetrazine caused by intramolecular repulsive N–N interaction plays a key role in accelerating the cycloaddition step. In contrast, we show that the observed reaction rates are way too high to be explained on this basis. It is widely accepted that the enhanced reactivity of these chemical tools is attributed to the electron-withdrawing effect of the heteroaryl substituent. Highly reactive 2-pyridyl-substituted tetrazines have become state of the art for time-critical processes and selective reactions at very low concentrations. The tetrazine/ trans-cyclooctene ligation stands out from the bioorthogonal toolbox due to its exceptional reaction kinetics, enabling multiple molecular technologies in vitro and in living systems.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
